Most people expect side effects from medications. Nausea from antibiotics, drowsiness from antihistamines, dizziness from blood pressure pills - these are common, predictable, and usually mild. But what about reactions that come out of nowhere? No warning. No pattern. Not even linked to how much you took? That’s where idiosyncratic drug reactions come in. These are the rare, mysterious, and sometimes deadly side effects that slip through every safety net we’ve built.
What Exactly Is an Idiosyncratic Drug Reaction?
An idiosyncratic drug reaction (IDR) isn’t just a bad reaction. It’s a reaction that makes no sense by normal rules. You take a drug at the right dose, for the right reason, and suddenly - days or weeks later - your body goes haywire. Your liver shuts down. Your skin starts peeling. Your lungs swell. And no one saw it coming.
Unlike the more common type A reactions - which are just exaggerated versions of a drug’s known effects - IDRs are type B. That means they’re unpredictable, not dose-dependent, and affect only a tiny fraction of users. The U.S. FDA reports these occur in roughly 1 in 10,000 to 1 in 100,000 people. Sounds rare? It is. But here’s the catch: while they make up only 13-15% of all adverse drug reactions, they’re responsible for over a third of all drug withdrawals from the market.
Think about it. Drugs go through years of testing, thousands of patients, and strict regulatory reviews. Yet, some reactions only show up after hundreds of thousands of people are taking the drug. That’s how sneaky these reactions are.
Why Do They Happen? The Science Behind the Mystery
No one fully understands why IDRs happen in some people and not others. But the leading theory? Your immune system gets tricked.
Some drugs break down in your liver into reactive chemicals - little molecular fragments that stick to your body’s own proteins. Your immune system doesn’t recognize these altered proteins. It sees them as invaders. So it attacks. That’s the hapten hypothesis. It’s why reactions like drug-induced liver injury (IDILI) or severe skin reactions like Stevens-Johnson syndrome (SJS) happen. Your body isn’t reacting to the drug. It’s reacting to the drug’s fingerprint on your cells.
Genetics play a huge role. Certain HLA gene variants - your body’s biological ID tags - make some people far more likely to have these reactions. For example:
- HLA-B*57:01 → Abacavir (HIV drug) → Severe hypersensitivity in 5-8% of carriers
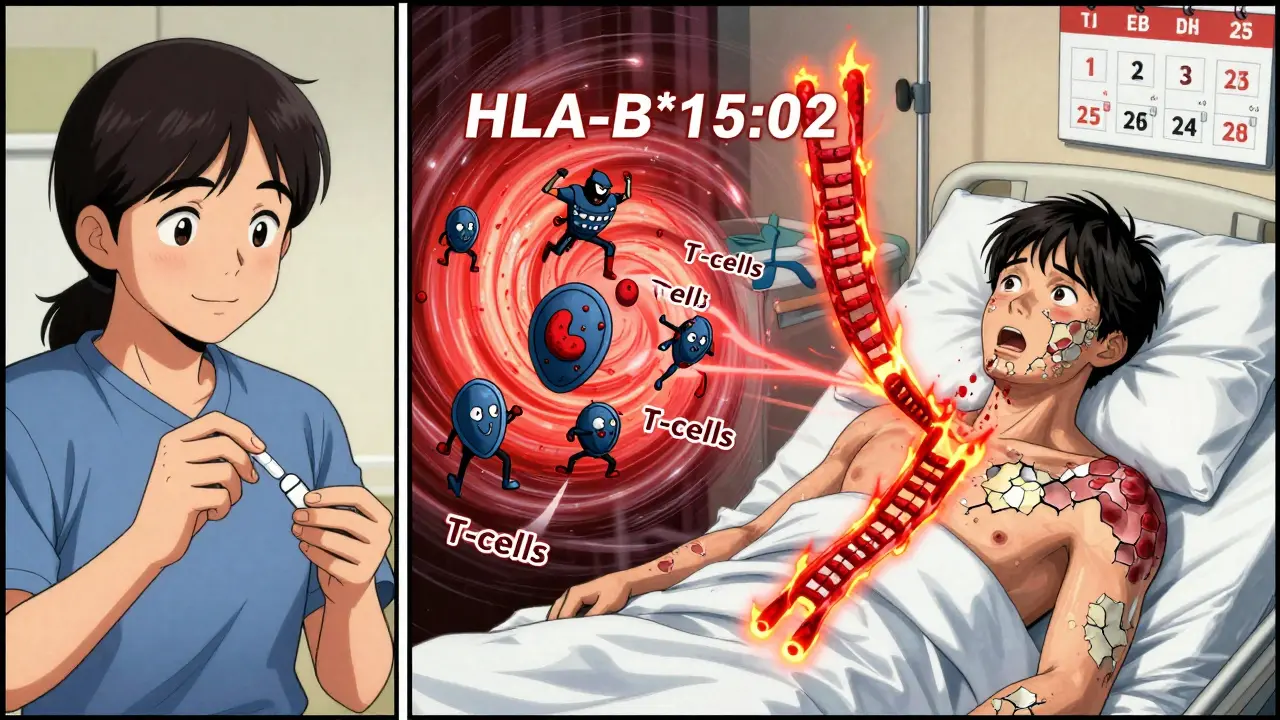
- HLA-B*15:02 → Carbamazepine (epilepsy drug) → 100x higher risk of SJS/TEN in Southeast Asians
- HLA-A*31:01 → Phenytoin → Strong link to skin reactions
These aren’t just correlations. We’ve tested them. The PREDICT-1 study showed that screening for HLA-B*57:01 before giving abacavir cut hypersensitivity cases from 8% to near zero. That’s the power of genetic testing - when it’s available.
But here’s the problem: we have validated genetic tests for fewer than 10 drugs out of thousands on the market. For the other 92% of IDRs, we have no way to predict who’s at risk.
Common Types of Idiosyncratic Reactions
Not all IDRs are the same. Some hit the liver. Others attack the skin. Here are the big three:
1. Drug-Induced Liver Injury (IDILI)
This is the most common serious IDR. It accounts for 45-50% of all severe drug-related liver damage. The injury can be:
- Hepatocellular (60-65%) - liver cells die off
- Cholestatic (30-35%) - bile flow blocked
- Mixed (5-10%) - both
Drugs like acetaminophen (in overdose), isoniazid, statins, and even some herbal supplements can trigger it. Symptoms? Fatigue, yellow skin, dark urine, nausea. But here’s the kicker: it often shows up 1-8 weeks after starting the drug. By then, doctors might think it’s hepatitis, flu, or even gallstones. The Roussel Uclaf Causality Assessment Method (RUCAM) is the gold standard tool doctors use to decide if the drug is to blame. A score above 8 means it’s "highly probable."
2. Severe Cutaneous Adverse Reactions (SCARs)
These are skin reactions so bad they can kill. Three main types:
- Stevens-Johnson Syndrome (SJS) - blisters and peeling on less than 10% of skin
- Toxic Epidermal Necrolysis (TEN) - peeling over 30% of skin, like a severe burn
- DRESS - drug reaction with eosinophilia and systemic symptoms - rash, fever, swollen lymph nodes, organ damage
Common culprits? Antibiotics (sulfa drugs), anticonvulsants (carbamazepine, phenytoin), allopurinol. The ALDEN algorithm helps doctors link the drug to the reaction. Mortality? Up to 35% for TEN. That’s higher than many cancers.
3. Other Organ Damage
IDRs can also affect the kidneys, lungs, heart, or blood. Think drug-induced lupus, vasculitis, or bone marrow failure. These are rarer but just as dangerous.
Why Are These Reactions So Hard to Spot?
Imagine you’re a doctor. A patient comes in with a rash, fever, and fatigue. They started a new medication three weeks ago. Is it the drug? A virus? A new allergy? A flare-up of their autoimmune disease?
You don’t have a blood test. No scan. No quick answer. You have to piece it together.
That’s the reality. Most IDRs:
- Appear after a delay (1-8 weeks)
- Look like other illnesses
- Don’t show up in lab tests until damage is advanced
- Are rarely reported - studies say up to 95% go undetected
A 2021 survey by the DRESS Syndrome Foundation found patients waited an average of 17.3 days for a correct diagnosis. Over 60% were initially told they had the flu or a viral infection. One patient described waiting a week with fever, rash, and trouble breathing before her HIV specialist finally said, "This might be abacavir."
And here’s the cruel twist: once you’ve had one IDR, you can’t take that drug again - ever. Even a tiny amount can trigger a worse reaction. No second chances.
How Are IDRs Diagnosed and Managed?
There’s no magic test. Diagnosis is detective work.
Step 1: Stop the drug. If symptoms improve within days, that’s a strong clue. This is called a "dechallenge."
Step 2: Rule out other causes. Infections? Autoimmune? Cancer? Liver function tests? Blood counts? Imaging?
Step 3: Use validated tools. RUCAM for liver injury. ALDEN for skin reactions. Lymphocyte transformation tests (LTT) - though only 60-70% accurate - can sometimes confirm immune involvement.
Step 4: Don’t rechallenge. Re-exposing someone to the drug to confirm the reaction? Almost never done. Too risky. Only in rare research settings.
Treatment? Stop the drug. Supportive care. Steroids for DRESS. ICU for TEN. Liver transplant for severe IDILI. Recovery can take months. Some patients are left with permanent organ damage.

The Hidden Cost: Patients, Doctors, and Drug Companies
These reactions don’t just hurt patients. They break the system.
Patients face:
- Long hospital stays (average 12.4 days)
- Chronic conditions (28% develop lasting liver or kidney damage)
- Average out-of-pocket cost: $47,500
- Feeling dismissed by doctors who don’t recognize the signs
Doctors struggle because:
- Only 42% of drug labels give clear IDR warnings
- It takes 18-24 months of focused experience to reliably diagnose IDRs
- Most medical schools barely teach them
Drug companies lose billions. The Tufts Center estimates IDR-related failures cost the industry $12.4 billion a year. Between 1950 and 2023, 38 drugs were pulled from the U.S. market because of IDRs - including troglitazone (liver failure), bromfenac (liver death), and cerivastatin (muscle breakdown).
Now, companies screen for reactive metabolites in early development. Pfizer and others set strict limits: less than 50 pmol/mg of protein in preclinical tests. But even that doesn’t catch everything.
What’s Changing? The Future of Prediction
There’s hope. And it’s coming fast.
- In 2023, the FDA approved the first predictive test for pazopanib liver toxicity - 82% accurate.
- New HLA associations have been found for over 17 drugs, including phenytoin and diclofenac.
- The NIH just invested $47.5 million into the Drug-Induced Injury Network to map out the biological pathways.
- Europe’s "ADRomics" project aims to predict IDRs using DNA, proteins, and metabolism data by 2027.
AI is helping too. Companies like ArisGlobal and Oracle Health are building algorithms that scan millions of patient records to find hidden patterns. But none have cracked above 70% accuracy yet.
The goal? Not to eliminate IDRs. That’s impossible. But to reduce them. Experts believe we can cut severe cases by 60-70% in the next 10 years - through better screening, smarter drug design, and genetic testing where it matters.
What Should You Do?
If you’re on medication:
- Know the common signs: unexplained rash, fever, dark urine, yellow eyes, extreme fatigue, swelling
- Track when symptoms started - did they appear 1-8 weeks after starting a new drug?
- Don’t ignore "odd" symptoms. If something feels wrong, speak up.
- If you’ve had one IDR, keep a list of all drugs you can’t take again - and share it with every doctor.
- Ask: "Is there a genetic test for this drug?" Especially if you’re of Southeast Asian, African, or Mediterranean descent.
If you’re a patient with a rare condition - like HIV, epilepsy, or autoimmune disease - you’re at higher risk. Stay informed. Use trusted resources like LiverTox (updated weekly) or RegiSCAR (updated quarterly). They’re free, detailed, and built by experts.
Idiosyncratic reactions are rare. But they’re real. And they’re getting better understood. The next time you hear about a drug being pulled from the market, don’t just think "safety failure." Think: "Someone’s body reacted in a way science still can’t fully explain." And that’s why we keep looking.
What is the difference between idiosyncratic and predictable drug reactions?
Predictable (type A) reactions are dose-related and happen because of how the drug works - like drowsiness from antihistamines or bleeding from blood thinners. They affect 80-85% of all adverse reactions. Idiosyncratic (type B) reactions are unrelated to the drug’s main effect, occur in rare individuals, aren’t tied to dose, and often involve the immune system. They’re unpredictable, hard to detect, and can be life-threatening.
Can you test for idiosyncratic drug reactions before taking a drug?
Only for a few drugs. Genetic tests exist for HLA-B*57:01 (abacavir), HLA-B*15:02 (carbamazepine), and HLA-A*31:01 (phenytoin). These are highly accurate at preventing reactions in people who carry the gene. But for the vast majority of drugs - over 90% - there are no reliable pre-treatment tests. That’s why IDRs remain unpredictable.
How long after starting a drug can an idiosyncratic reaction occur?
Most appear between 1 and 8 weeks after starting the drug. Liver injuries often take 2-6 weeks. Skin reactions like DRESS or SJS usually show up around 2-4 weeks. But some cases have been reported as early as 3 days or as late as 12 weeks. This delay is why doctors often miss the connection.
Are idiosyncratic reactions more dangerous than common side effects?
Yes - in terms of severity. While common side effects are usually mild and temporary, IDRs can cause permanent organ damage, require hospitalization, or be fatal. Up to 35% of toxic epidermal necrolysis cases are deadly. IDRs are responsible for 30-40% of all drug withdrawals from the market, even though they affect far fewer people than common side effects.
Can you get an idiosyncratic reaction from a drug you’ve taken before?
Yes. The first exposure might not trigger a reaction. But once your immune system has been sensitized - even without symptoms - a second exposure can cause a severe reaction. This is why doctors advise never re-taking a drug that caused a suspected IDR, even if it was mild the first time.
What should I do if I think I’m having an idiosyncratic reaction?
Stop the drug immediately and contact your doctor. Don’t wait. If you have a rash with fever, blistering skin, yellowing eyes, or severe fatigue, go to an emergency department. Bring a list of all medications you’ve taken in the last 8 weeks. Ask if it could be a drug-induced reaction. Early recognition saves lives.
14 Comments
Write a comment
More Articles

Antiseizure Medications and Generic Substitution: Risks and Best Practices
Generic antiseizure medications can save money, but for narrow therapeutic index drugs like lamotrigine and carbamazepine, even small changes in absorption can trigger seizures. Learn the risks, who’s most vulnerable, and how to protect yourself.

Understanding the Different Types of Headaches: A Complete Guide
A comprehensive guide that explains every major headache type, symptoms, triggers, and treatment options so you can manage pain and know when to seek help.

Top Alternatives to FelixForYou.ca for Telemedicine in 2025
Discover the leading alternatives to FelixForYou.ca, a Canadian telemedicine platform, focusing on digital healthcare options available in 2025. Highlighting Maple Health, Tia Health, Livewell, Telus Health, and Jill Health, this article provides insights into each service's offerings, pros, and cons. Whether looking for 24/7 access to healthcare professionals, adaptable programs, or specialized medical services, these alternatives cater to varied health needs. Consider factors such as accessibility, cost, and service range to choose the right telemedicine solution.
Stephanie Paluch
March 12, 2026 AT 22:50I had a friend who got DRESS from allopurinol. She was fine one week, then woke up with a full-body rash and fever. Took 3 months to recover. Never thought a gout pill could do that. 😭